REMDESEVIR IN ADULTS WITH SEVERE COVID-19: A RANDOMISED, DOUBLE-BLIND, PLACEBO CONTROLLED, MULTICENTRE TRIAL
Yeming Wang*, Dingyu Zhang*, Guanhua Du*, Ronghui Du*, Jianping Zhao*, Yang Jin*, Shouzhi Fu*, Ling Gao*, Zhenshun Cheng*, Qiaofa Lu*, Yi Hu*, Guangwei Luo*, Ke Wang, Yang Lu, Huadong Li, Shuzhen Wang, Shunan Ruan, Chengqing Yang, Chunlin Mei, Yi Wang, Dan Ding, Feng Wu, Xin Tang, Xianzhi Ye, Yingchun Ye, Bing Liu, Jie Yang, Wen Yin, Aili Wang, Guohui Fan, Fei Zhou, Zhibo Liu, Xiaoying Gu, Jiuyang Xu, Lianhan Shang, Yi Zhang, Lianjun Cao, Tingting Guo, Yan Wan, Hong Qin, Yushen Jiang, Thomas Jaki, Frederick G Hayden, Peter W Horby, Bin Cao, Chen Wang
29 NİSAN 2020: THE LANCET, https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(20)31022-9/fulltext
Background
No specific antiviral drug has been proven effective for treatment of patients with severe coronavirus disease 2019 (COVID-19). Remdesivir (GS-5734), a nucleoside analogue prodrug, has inhibitory effects on pathogenic animal and human coronaviruses, including severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in vitro, and inhibits Middle East respiratory syndrome coronavirus, SARS-CoV-1, and SARS-CoV-2 replication in animal models.
Methods
We did a randomised, double-blind, placebo-controlled, multicentre trial at ten hospitals in Hubei, China. Eligible patients were adults (aged ≥18 years) admitted to hospital with laboratory-confirmed SARS-CoV-2 infection, with an interval from symptom onset to enrolment of 12 days or less, oxygen saturation of 94% or less on room air or a ratio of arterial oxygen partial pressure to fractional inspired oxygen of 300 mm Hg or less, and radiologically confirmed pneumonia. Patients were randomly assigned in a 2:1 ratio to intravenous remdesivir (200 mg on day 1 followed by 100 mg on days 2–10 in single daily infusions) or the same volume of placebo infusions for 10 days. Patients were permitted concomitant use of lopinavir–ritonavir, interferons, and corticosteroids. The primary endpoint was time to clinical improvement up to day 28, defined as the time (in days) from randomisation to the point of a decline of two levels on a six-point ordinal scale of clinical status (from 1=discharged to 6=death) or discharged alive from hospital, whichever came first. Primary analysis was done in the intention-to-treat (ITT) population and safety analysis was done in all patients who started their assigned treatment. This trial is registered with ClinicalTrials.gov, NCT04257656.
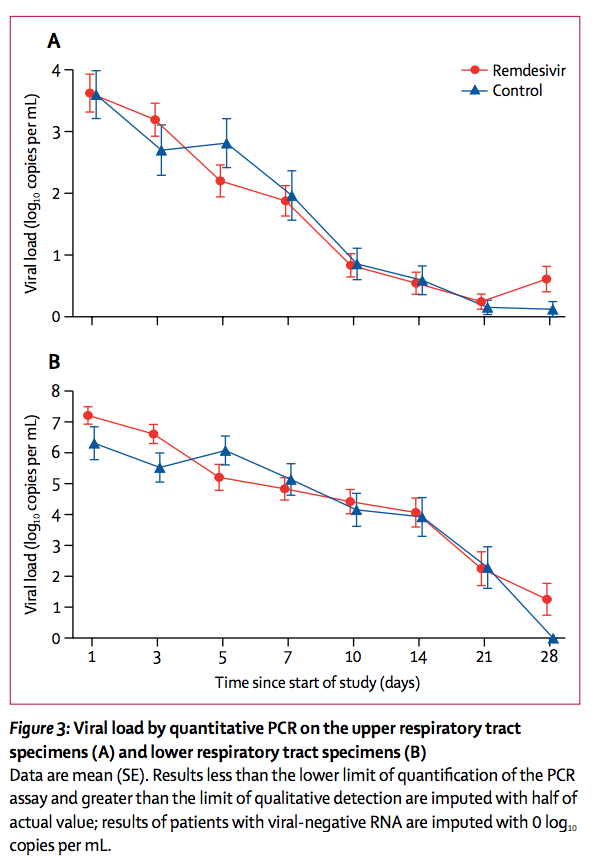
Findings
Between Feb 6, 2020, and March 12, 2020, 237 patients were enrolled and randomly assigned to a treatment group (158 to remdesivir and 79 to placebo); one patient in the placebo group who withdrew after randomisation was not included in the ITT population. Remdesivir use was not associated with a difference in time to clinical improvement (hazard ratio 1·23 [95% CI 0·87–1·75]). Although not statistically significant, patients receiving remdesivir had a numerically faster time to clinical improvement than those receiving placebo among patients with symptom duration of 10 days or less (hazard ratio 1·52 [0·95–2·43]). Adverse events were reported in 102 (66%) of 155 remdesivir recipients versus 50 (64%) of 78 placebo recipients. Remdesivir was stopped early because of adverse events in 18 (12%) patients versus four (5%) patients who stopped placebo early.
Interpretation
In this study of adult patients admitted to hospital for severe COVID-19, remdesivir was not associated with statistically significant clinical benefits. However, the numerical reduction in time to clinical improvement in those treated earlier requires confirmation in larger studies.
FULL MAKALE İLİŞİKTEDİR







